As a transplant survivor and peer volunteer, I have met with over 150 transplant patients. The most common question I hear concerns what recovery looks like. People want to know about timelines, precautions, complications, medications, benchmarks, and much more.
I can only answer these questions from my experience, and no two outcomes are the same. But I’ve read and heard enough other stories to know where mine is typical or exceptional, so I can also place my story in a broader context.
In June of 2016, I was diagnosed with acute myeloid leukemia. I underwent induction chemotherapy and achieved a temporary remission. In October of 2016, I received a double cord blood stem cell transplant. I fully recovered and have returned to all my prior activities, so mine is a very positive story. Along the way, however, there were several memorable challenges.
Prelude to a transplant
My initial treatment required a five-and-a-half-week hospital stay. It was one week for the traditional “7+3” chemotherapy regimen, and another four and a half weeks to monitor and treat the inevitable infections that followed in the wake of chemotherapy-induced immunosuppression.
My diagnosis was routine for my providers but shocking for me. I was asymptomatic and feeling perfectly healthy at my annual physical. But low white blood cell counts triggered a bone marrow biopsy that established my diagnosis. I was hospitalized the next day and started chemotherapy the day after that. My treatment was underway before I even understood my disease and its bleak prognosis.
When they told me to expect a 5-6-week hospital stay, I was dumbfounded. I quickly realized that I needed ways to cope with how my world had suddenly become very small and quite precarious. Over the ensuing weeks, I developed and honed several crucial strategies.
First, I relied upon mindfulness, meditation, and yoga. It helped me banish thoughts about the past and anxieties about the future, and to non-judgmentally accept and live in each moment as it unfolded.
Second, I did as much physical exercise as my circumstances would allow. My routines included stretching, isometric exercises, extensive hall walking, squats, lunges and pushups. I did it mindfully, and these routines structured my days, increased my energy, and lifted my spirits.
Third, I was a pro-active patient. I cultivated good communication with my doctors and nurses. I asked lots of questions about my treatment and became a collaborator in decisions about medications, dosing, and deciphering and treating the many infections and side effects that came my way.
Fourth, I maintained my robust sense of humor. Sharing jokes and witty banter with my medical providers broke the ice, resolved the tension, and humanized our consults. It also gave friends and family a way to relate to me as the person I’d always been rather than the patient I’d recently become.
Fifth, I relied on a supportive belief system. For some, that’s religion. For me, it was a secular worldview based on my social science background. It encouraged me to learn about my condition and fostered a practical, problem-solving orientation to all the challenges it posed.
Finally, I wrote my story from the very first week. I sent detailed reports about my status and reflections as a cancer patient to a large group of email correspondents. Writing for others forced me to understand my journey so I could articulate it for them. This writing became a psychic survival mechanism (and a subsequent memoir).
When the time for transplant arrived, I packed a bag, grabbed my laptop, and took these coping strategies with me. As doctors cured my body, these strategies sustained me throughout everything that was to come.
The Transplant and Hospitalization
Like many patients, I was admitted to my transplant hospital one week before the actual procedure (day -7). I underwent conditioning chemotherapy and full body radiation. Upon my transplant (day 0), I was told to expect another three to five weeks in the hospital before I could be safely discharged.
Days 1-7 were uneventful except for some moderate nausea due my prior chemotherapy and radiation. I got some relief from a drug called marinol that allowed me to eat regular meals during this time. As my counts hit bottom, I was closely monitored for fevers and infections. Even so, I felt good enough to do daily exercise, walk on a treadmill, do yoga, and be as active as possible while confined to my room.
On day 8, my doctors said I was doing so well they were thinking of discharging me in a couple more days – much earlier than expected. But then I developed an infection and a recurring fever that spiked every twelve hours for several days and delayed my discharge.
By day 19, my infection and fevers had resolved, and I went home under the watchful eye of my caregivers. I thus left the hospital in just under three weeks since transplant – much quicker than the 3-5-week hospitalization I had been told to expect.
A Memorable Month at Home
From day 20 to 50, the plan was for daily clinic visits to monitor counts, treat symptoms, and assess progress. On day 21, a bone marrow biopsy revealed that one of my donors was 99% engrafted, which was an unusually early and complete success for a cord blood transplant. With engraftment underway, we then watched for signs of graft-vs.-host disease.
During this month (day 20-50), my caregivers were essential. They drove me to daily clinic visits for blood draws, provider consultations and needed treatments. From day 20-26, I received daily transfusions of platelets and several transfusions of red blood cells. Several more transfusions as well as injections of growth factor medications to spur new white blood cells followed throughout this month. After the second week, however, they reduced my clinic visits to fewer and fewer days each succeeding week.
That first month at home (day 20-50) was also when I felt the side-effects from my treatment most keenly. The lingering impacts of chemotherapy and radiation, the engraftment process, and multiple medications produced several memorable symptoms. There were aches and pains from the engraftment itself that I treated with ibuprofen, and ongoing bouts of nausea that I managed with marinol. I was also taking about 20 scheduled pills a day, including prophylactic antibiotics, anti-viral and anti-fungal medications, anti-rejection medications, and several pills to manage side effects of these medications.
My most memorable symptom during this period was a staggering level of fatigue as my body underwent this transformation. I was sleeping eight to nine hours a night but still required lengthy naps in the late morning and late afternoon. I couldn’t stay awake for more than four hours at a time and was totally exhausted by nightfall.
On day 39, routine blood work detected a cytomegalovirus infection. It’s one of many critters that can reside in our gut our whole lives unbeknownst to us. But with immunosuppression, the virus can become active and pose serious danger. It is usually well controlled if detected early and treated quickly, so I was immediately put on a more powerful antiviral drug to address the infection.
The virus drastically reduced my white blood cell count while the antiviral medication added further immunosuppressive effects. For a few days, I had additional fatigue, aches, chills, and nausea. When the virus and anti-viral forces fought to a standstill, they contemplated admitting me back into the hospital for several days of IV, antiviral treatments. Instead of re-hospitalization, however, the compromise treatment was an outpatient infusion of IV immunoglobulin to boost my white blood cell count while the antiviral medication gradually tamed the virus. With that, I continued my recovery at home.
Through the First 100 Days
From day 50 to day 100, I experienced gradual if uneven improvement. Clinic visits tapered to once a week or less. Bone aches ceased and nausea all but disappeared. Fatigue also decreased, and when I did feel tired, I could usually trace it to increased activity levels compared to my first 30 days at home. As I was able to reduce doses or eliminate some medications, my mind cleared and my energy increased. While I experienced minor rashes, dry eyes, and sinus headaches, there was nothing that required major medical treatment or raised suspicions of graft-vs.-host disease.
By day 58, I began experiencing neuropathy in my feet. This is a common side-effect of chemotherapy, but in my case, it has been blessedly mild. It mainly presents as numbness and tingling under the balls of both feet. I was told it might resolve within a year, but it remains the only side effect that has persisted and which I now regard as permanent. It has not responded to acupuncture or cortisone injections. My best adaptation has been specially designed shoes and custom insoles that take pressure off the sensitive areas and make the condition quite tolerable.
By day 60, I was having trouble lining up caregivers but still needed to get to weekly clinic visits. I had been prohibited from driving or being without a caregiver for the first 100 days, but that was no longer practical. I carefully began driving myself to clinic visits. By then, I knew how my medications affected me and so I delayed my antifungal medication and the blurry vision it caused until I safely returned from my outings.
On day 78, my oncologist recommended removing the “Power Hickman” central line that had served me well for almost seven months. It had been with me since the beginning of my treatment and had facilitated painless blood draws and countless infusions of blood, platelets, IV medications, and chemotherapy. But with the reduction in all these procedures, the risk of an infected line was becoming greater than the benefits of keeping it in place. An added benefit was being able to take a shower without wrapping my entire upper torso in Saran Wrap to protect the gizmo.
Day 100 was a significant benchmark for several reasons. I had another bone marrow biopsy that confirmed full engraftment and no residual leukemia. Reviewing my biopsy results, blood tests, and overall progress, my oncologist said my recovery to date was “as good as it gets.”
At this time, I was able to eliminate or reduce many of my medications. More importantly, I began to gradually taper my anti-rejection medication (cyclosporine) over the next three-month period. The gradual pace of this taper was meant to allow my old body and my new immune system to learn to get along with each other, restore full immunity, and avoid GVHD
By this time, I was feeling much better and was eager to return to my regular activities. Since my blood counts were all good, I asked my oncologist her advice. She provided a rather technical explanation of why I was still at considerable risk and needed to avoid crowds, continue wearing my mask in public, and follow other precautions.
My layman’s interpretation of her explanation was that even though I had sufficient white blood cells and neutrophils, my anti-rejection medication would still prevent them from fully activating in case of infection. So despite feeling better and having good counts, I needed to maintain precautions until my anti-rejection medication had run its course and my immune system was more functional and able to protect me in a germ-filled world.
Completing the Marathon
From day 100 to day 180, I continued gradual improvement and weathered some minor bumps in the road. My clinic visits were now spaced out every couple weeks, and I began to see other practitioners to assess some peripheral issues arising from my diagnosis and treatment.
Since my leukemia put me at risk for skin cancer, I saw a dermatologist who detected a small, basal cell carcinoma that was easily excised. I continue to see her every six months for full body skin checks with no further issues. My leukemia had also caused some retinal hemorrhaging that was diagnosed before transplant. A follow up visit during this period showed that all retinal issues had completely resolved with the eradication of my leukemia.
Even though I was now tapering my anti-rejection medication, its cumulative impact produced numerous unpleasant side effects. While I avoided the most serious ones, I nonetheless experienced flushing, hypertension, nausea, altered kidney function, neuropathy, weight loss, leg cramps, sinus irritation, abdominal swelling, and night sweats. I began a temporary regimen of blood pressure medication and rode out the other issues. To top it off, I also had a flare up of the cytomegalovirus, which once again was quickly detected and effectively treated with specialized antiviral medication.
On day 180, I had my 6-month biopsy which reconfirmed full engraftment and no residual leukemia. At this time, I stopped my anti-rejection medication and its unwanted side effects began to dissipate. I was also able to stop virtually all of my remaining pills with the exception of an antiviral medication which continued until day 365. With adequate immunity restored, I was cleared to do any activity I wanted with one exception: I had to avoid fungal sources of infection (yard work, turning over soil, fresh mushrooms, etc.) for the next six months because such infections are easy to contract and difficult to eradicate.
For me, this was a major psychological turning point. I accepted that I was actually better, resumed my “normal” life, and let go of lingering anxieties about my status. When my transplant oncologist said she didn’t need to see me for another six months, it was initially unnerving after such intensive monitoring. At the same time, it reinforced my sense that I had reached a major milestone in my recovery.
“As Good As It Gets” (and Some Cheap Advice)
After day 180, my care shifted back to my initial oncologist at my induction hospital. Monthly blood draws and bimonthly consultations gradually became less frequent. Four years out from my initial diagnosis, I now have blood draws four times a year and see this oncologist twice a year.
At year one and two (days 365 and 730), I returned to my transplant oncologist for my final two biopsies which found no residual disease. At year one, they re-did my childhood vaccinations from dead viral sources; at year two, I received my remaining vaccinations from live viral sources.
There’s good reason to say my story is “as good as it gets.” First, I got into remission on the first round of induction chemotherapy. This does not happen for a significant minority of AML patients who require multiple rounds of chemotherapy or other treatments to attain remission.
Second, I had full donor engraftment in three weeks. Most patients achieve engraftment, but it typically takes longer or doesn’t happen as completely as it did in my case. In the worst-case scenario, a small percentage of patients never experience engraftment and face a very poor prognosis.
Third, I have had no graft-vs.-host disease. I had been told there was a 60-70% chance of acute (within the first 100 days) GVHD in cases like mine, but I had no symptoms that could be attributed to this cause. That reduced my chances of chronic (after the first 100 days) GVHD to 20%. Although it can appear years after transplant, I’ve had no symptoms as of this writing.
What is typical about my story are the various infections, unpleasant side-effects, and minor complications documented here. They are simply part and parcel of the disease, treatment, and transplant; few if any patients escape them altogether. But in my case, they were quite manageable with the excellent support I received from my medical practitioners and caregiver team. Thanks to them, I left my transplant hospital on day 19 and never returned.
Advice is cheap, so here’s my two cent’s worth. Even in the best-case scenario, recovery is so gradual that it’s hard to realize when you are actually making progress (especially when there are periodic setbacks). I learned to pay attention to even small steps of improvement and took heart when they occurred.
Here’s one example. Around day 40, I ran up a flight of stairs at home and became short of breath. I initially found this discouraging, but then I realized I hadn’t even run up a flight of stairs since my diagnosis, and that this was progress not regress. Recovery happens through small, incremental changes that eventually culminate in qualitative improvement. It helps to be aware of these small steps as they occur; you may even want to record them in a weekly journal to fully appreciate them.
Finally, some clichés bear repeating. Recovery is a marathon, not a sprint. Moreover, it’s a marathon on an obstacle course of potential complications. Don’t hesitate to ask for help from your doctors or accept assistance from your caregivers. It’s not a burden; it actually makes them feel better when they can help you out. Finally, cultivate patience, resilience, and fortitude as you go the distance. It will serve you well.
Steve Buechler is the author of “How Steve Became Ralph: A Cancer/Stem Cell Odyssey (with Jokes),” which was recently selected as an inspirational “Suggested Reading” by the Leukemia and Lymphoma Society. He recently recorded a 5-part webcast series that expands on his recovery story here. Steve’s numerous blog posts, video presentations, and more may be found at www.stevebuechlerauthor.com and he is on Facebook at SteveBuechlerAuthor.




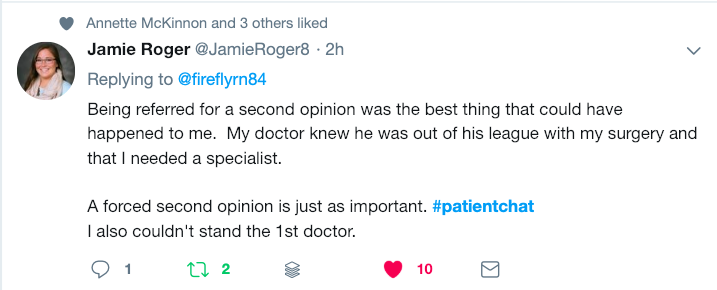
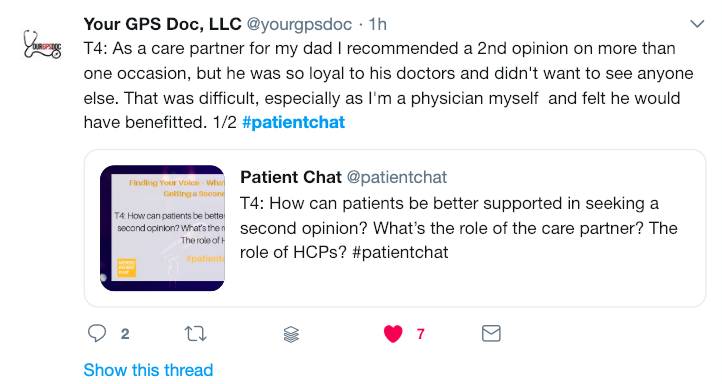

 Have you ever had the experience of leaving the doctor’s office wishing you had remembered to ask a certain question? Or have you left it until the very end to tell your doctor about the real reason for your visit? These so-called “doorknob” questions – bringing up an important concern just as you are leaving the office – can mean your doctor won’t have time to adequately address your concerns. When the average time it takes for a doctor’s visit is fifteen minutes, it’s easy to feel rushed and forget what you wanted to say, or to leave an appointment unsure of the information you have heard. But with a little advance preparation you can learn how to make the most of those fifteen minutes. Follow these fifteen tips to become a more empowered and engaged partner in your own health – and the health of those you care for.
Have you ever had the experience of leaving the doctor’s office wishing you had remembered to ask a certain question? Or have you left it until the very end to tell your doctor about the real reason for your visit? These so-called “doorknob” questions – bringing up an important concern just as you are leaving the office – can mean your doctor won’t have time to adequately address your concerns. When the average time it takes for a doctor’s visit is fifteen minutes, it’s easy to feel rushed and forget what you wanted to say, or to leave an appointment unsure of the information you have heard. But with a little advance preparation you can learn how to make the most of those fifteen minutes. Follow these fifteen tips to become a more empowered and engaged partner in your own health – and the health of those you care for.


