How to Read and Understand a Scientific Paper
In a previous article, How to Read Beyond the Headline: 9 Essential Questions to Evaluate Medical News, I recommended you should always try to read an original study (if cited) to evaluate the information presented. In this follow-on article, you will learn how to read a scientific research paper so that you can come to an informed opinion on the latest research in your field of interest. Understanding research literature is an important skill for patient advocates, and as with any skill, it can be learned with practice and time.
Let’s start by looking at what exactly we mean by the term “scientific paper”. Scientific papers are written reports describing original research findings. They are published in peer reviewed journals, which means they have been refereed by at least two other experts (unpaid and anonymized) in the field of study in order to determine the article’s scientific validity.
You may also come across the following types of scientific papers in the course of your research.
• Scientific review papers are also published in peer reviewed journals, but seek to synthesize and summarize the work of a particular sub-field, rather than report on new results.
• Conference proceedings, which may be published in a journal, are referred to as the “Proceedings of Conference X”. They will sometimes go through peer review, but not always.
• Editorials, commentaries and letters to the editor offer a review or critique of original articles. They are not peer-reviewed.
Most scientific journals follow the IMRD format, meaning its publications will usually consist of an Abstract followed by:
• Introduction
• Methods
• Results
• Discussion
Let’s look at each of these sections in turn.
(a) Introduction
The Introduction should provide you with enough information to understand the article. It should establish the scientific significance of the study and demonstrate a relevant context for the current study. The scope and objectives of the study should be clearly stated.
When reading the Introduction, ask yourself the following questions:
· What specific problem does this research address?
· Why is this study important?
(b) Methods
The Methods section outlines how the work was done to answer the study’s hypothesis. It should explain new methodology in detail and types of data recorded.
As you read this section, look for answers to the following questions:
- What procedures were followed?
- Are the treatments clearly described?
- How many people did the research study include? In general, the larger a study the more you can trust its results. Small studies may miss important differences because they lack statistical power. Case studies (i.e. those based on single patients or single observations) are no longer regarded as scientific rigorous.
- Did the study include a control group? A control group allows researchers to compare outcomes in those who receive a treatment with those who don’t.
(c) Results
The Results section presents the study’s findings. It should follow a logical sequence to answer the study hypothesis. Pay careful attention to any data sets shown in graphs, tables, and diagrams. Try to interpret the data first before reading the captions and details. If you are unfamiliar with statistics, you will find a helpful glossary of terms here. Click here for an online guide to help you understand key concepts of statistics and how these concepts relate to the scientific method and research.
Consider the following questions:
- Are the findings supported by persuasive evidence?
- Is there an alternative way to interpret these findings?
(d) Discussion
The Discussion places the study in the context of the broader field of research. It should explain how the research has moved the body of scientific knowledge forward and outline the next steps for further study.
Questions to ask:
• Does the study have any limitations? Limitations are the conditions or influences that cannot be controlled by the researcher. Any limitations that might influence the results should be mentioned in the study’s findings.
- How are the findings new or supportive of other work in the field?
- What are some of the specific applications of the study’s findings?
The IMRD format provides you with a useful framework to read a scientific paper. You will need to read a paper several times to understand its findings. Consider your first reading of the study as a “big picture” reading. Scan the Abstract for a summary of the study’s principal objectives, the methods it used and the principal conclusions. A well-written abstract should allow you to identify the basic content of an article to determine its relevance to you. In describing how she determines the relevance of a study, research RN, Katy Hanlon, focuses on “key words and phrases first. Those that relate to the author/s base proposal as well as my own interests”. Medical writer, Nora Cutcliffe, also scans upfront “to gauge power and relevance of clinical trial data”. She looks for “study enrollment (n), country and year”. It’s important to note the publication date to determine if this article contains the latest findings or if there is more up-to-date research available. Cutcliffe also advises you should “note author affiliations and study sponsors”. Here you are looking out for any potential bias or vested interest in a particular outcome. Check the Acknowledgments section to see if the author(s) declare any financial interests in the research which might bias their findings. Finally, check if the article is published in a credible journal. You will find reputable biomedical journals indexed by Pubmed and Web of Science.
Next, circle or take note of any scientific terms or keywords you don’t understand and look up their meaning before your second reading. Scan the References section – you may even want to read an article listed here first to help you better understand the current study.
With the second reading you are going to deepen your comprehension of the study. You’ll want to highlight key points, consult the references, and take notes as you read. According to the scientific publisher, Elsevier, “reading a scientific paper should not be done in a linear way (from beginning to end); instead, it should be done strategically and with a critical mindset, questioning your understanding and the findings.” Scientist, Dr Jennifer Raff, agrees. “When I’m choosing papers to read, I decide what’s relevant to my interests based on a combination of the title and abstract”, she writes in How to read and understand a scientific paper: a guide for non-scientists. “But when I’ve got a collection of papers assembled for deep reading, I always read the abstract last”. Raff explains she does this “because abstracts contain a succinct summary of the entire paper, and I’m concerned about inadvertently becoming biased by the authors’ interpretation of the results”.
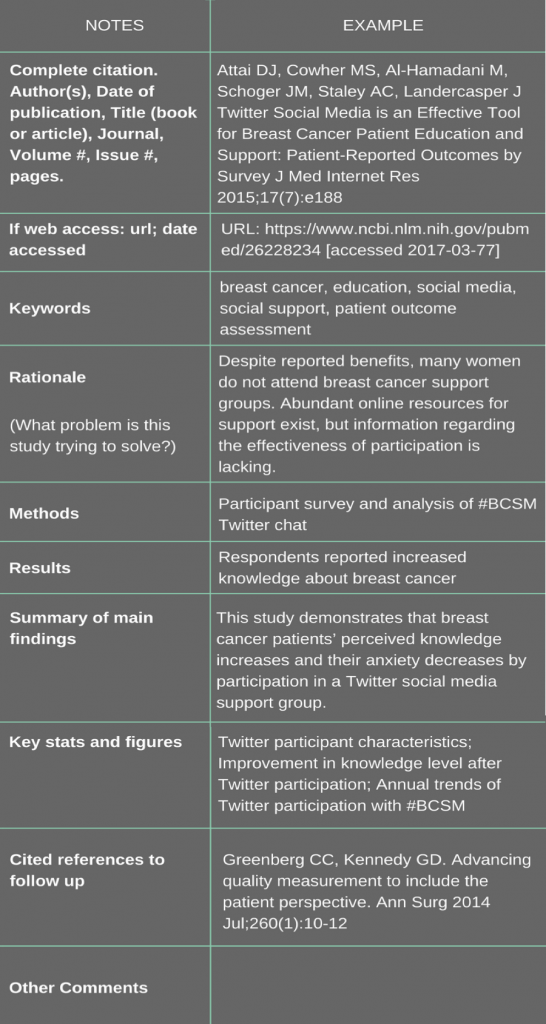
When you have read the article through several times, try to distill it down to its scientific essence, using your own words. Write down the key points you have gleaned from your reading such as the purpose of the study, main findings and conclusions. You might find it helpful to develop a template for recording notes, or adapt the template below for use. You will then have a useful resource to find the correct reference and to cross reference when you want to consult an article in the future.
In the example below I have taken an article published in 2015, as an example. You can read the paper Twitter Social Media is an Effective Tool for Breast Cancer Patient Education and Support: Patient-Reported Outcomes by Survey on PubMed.
Template for Taking Notes on Research Articles
Further reading
- How to read and review a scientific journal article. Duke University Writing Studio
- How to read a scientific paper. John W. Little & Roy Parker–University of Arizona

A Stanford Medicine X e-Patient scholar, Marie Ennis O’Connor is an internationally recognized keynote speaker, writer, and consultant on global trends in patient engagement, digital health and participatory medicine. Marie’s work is informed by her passion for embedding the patient voice at the heart of healthcare values. She writes about the experience of transitioning from breast cancer patient to advocate on her award-winning blog Journeying Beyond Breast Cancer.










