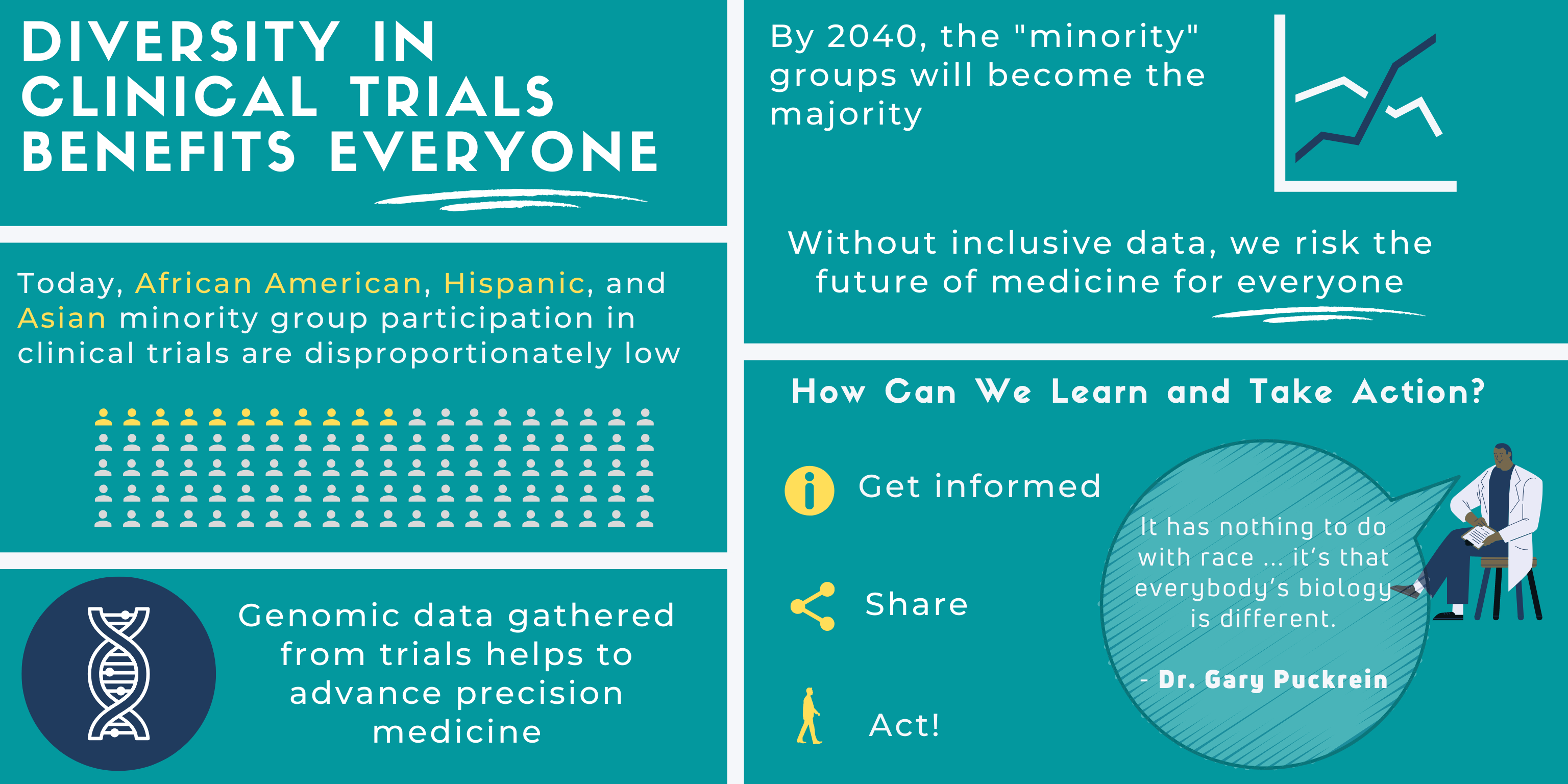
Diversity in Clinical Trials Benefits Everyone
The COVID-19 pandemic has been an unprecedented and collective experience for us all. Sadly, the pandemic has affected some more than others. This health crisis highlights both the pre-existing inequities in our healthcare system and the populations that experience such ramifications. Racial and ethnic minority groups (REMGs) will make up the majority of the population in the near future. Over the next two decades, the US population is estimated to make this major demographic shift. This is problematic because present inequities render REMGs disproportionately vulnerable and sick.
It is critical that REMGs are included in clinical trials because, as the broader population, their data will affect the outcome of precision medicine for everyone. Currently, REMGs do not have equitable access to general health care, let alone clinical trials. We need broader genomic data for understanding and curing existing diseases. Collective action is needed so future generations avoid the same health disparities.
Clinical Trials Awareness Needs to Extend Beyond an Awareness Week
Clinical trials awareness needs to extend beyond an awareness week or event. Diverse Health Hub had the opportunity to speak with Dr. Gary Puckrein, president of the National Minority Quality Forum, to learn more about the importance of diversity in clinical trials. In our one-on-one, we learn what’s at stake and what solutions we can act on collectively.
Dr. Puckrein shares three critical considerations. First, we must identify the barriers that prevent diverse clinical trial participation. Second, he describes the bleak outlook if we do not increase diversity in clinical trials. Lastly, we discuss the path to empowerment that creates solutions to these problems.
Historically, clinical trials have been flawed. Different types of barriers prevent diverse participation. For example, in the 1990s, women were not recruited into trials on the assumption that they would react to medications the same way men did:
“Back in the 1990’s we didn’t recruit women into clinical trials and so there was a big struggle to make sure that clinical trials were diverse by gender. Back then the assumption was if you have a bunch of men in the trial, women would react the same way that men do to the medications or the disease pathology would be the same and so, why would you need to have women in trials?”
Likewise, during that time, REMG participation rates were low, around 3-5%. Trust is an essential barrier, with a negative historical context. A poignant example of this for African Americans is the Tuskegee experiment. Dr. Puckrein explains:
“In the African-American community, there are fears that came out of the Tuskegee experiment where therapy was withheld from African-Americans who were suffering from syphilis for no particular reason and it has an awful impact on their lives and so, that’s a deep scar in the African-American community about participating in clinical trials.”
Mistrust in Clinical Trials is Systemic
Native American and Hispanic American communities have their own stories of mistrust in clinical trials.
The preexisting inequities in our healthcare system is another barrier, and a systemic one. Statistically, most REMGs don’t have access to basic healthcare, let alone resources to participate in clinical trials. Dr. Puckrein shares staggering data, “before the Affordable Care Act about 48% of African-Americans had no health insurance, about 54% of Hispanics had no health insurance.”
Furthermore, someone may be considered ineligible for a clinical trial if they already have existing health conditions. According to Dr. Puckrein, “REMGs were disproportionately excluded from some clinical trials based on either prior cancers, comorbidities, other things that may not have clinical implications.”
Outcome Risks If Barriers Aren’t Eliminated?
Without diversity in clinical trials, we risk collecting homogenized data which does not include the larger genomic context necessary to understand and improve precision medicine for everyone. It’s important that we make clinical trials all-inclusive, as Dr. Puckrein points out:
“What we’re finding is that we need highly targeted medicine, each individual is different. It has nothing to do with race or anything like that – it’s that everybody’s biology is different.”
The stakes are high because the future of our health depends on inclusivity right now. Clinical trials awareness needs to extend beyond a month of awareness. We don’t want today’s diseases to adversely affect generations to come. What then is a viable solution to this dilemma?
How Can We Learn and Take Action?
First. Get informed. Understand the larger historical context and intentions of clinical trials.
Second. Share this information by creating learning communities. Dr. Puckrein gives an example:
“If you bring physicians and patient advocacy groups, the policy makers and others around the table, you can solve problems like that and solve them quickly and inform the healthcare system so it’s interactive, it’s constantly learning. And so, we encourage people to build those – make them data driven. Use the data to help guide you on what interventions will work and how you’ll get the best possible outcome for patients.”
Third. Act. If you want to explore options in your treatment, seek out resources that embrace diversity in clinical trials. The “All of Us” Program is a public health initiative designed to remove the barriers that prevent inclusive access. As Dr. Puckrein highlights,
“The “All of Us” program is really designed to address that problem, to take populations that have not historically been in clinical trials — and now we include senior populations in that list as well — and make sure that we’re gaining signs in research about those populations. And so, the “All of Us” program is really designed to fill a big scientific gap in our knowledge.”
Furthermore, it is crucial to engage early with community leaders and grassroots organizations that resonate with underrepresented patients, and to encourage their participation in the critical stages of research.
Seeking knowledge, joining a community of advocates, and taking collective action is key to making a difference for all of us as we journey the path to empowerment.

Diverse Health Hub is a health equity education and awareness channel committed to spotlighting health disparities impacting vulnerable populations. DHH develops resources that forge needle-moving change to improve outcomes and create more equitable spaces for all patients.



